The rocky Road to an Adverse Event Report
Why just a small fraction of all adverse events is reported
This is the second article of a three-part series. However, this one is completely self-contained and is an informative read by itself. However, if you would like to learn more about my findings from Covid-vaccine-related VAERS analyses, please find the other articles here:
The rocky Road to an Adverse Event Report (this one)
The real Story behind the 'Bad Batches' (to come)
What is an Adverse Event?
Every day people get sick or die, so will some who have just been vaccinated. Vaccination is not necessarily the reason of sickness or death, so it is important to differentiate between an Adverse Event (timely correlation) and a side effect (causing correlation). A single adverse event is just „anecdotal“ evidence for a side effect but not proof. Though, a larger number of adverse events of a certain type is an important indicator for side effects that have not yet been detected. An adverse event reporting system is the only reasonable way to find rare side effects for which the sample size of clinical studies is too small.
In my personal opinion, each and every adverse effect that happens a short time after vaccination should be reported in order to assess its statistical relevance. If the number of adverse events of a certain type is significantly higher than its statistical expectation for a certain age group and gender, it is evidence for a side effect and needs to be verified with other measures. This way, it was finally proven and accepted that myocarditis and pericarditis are side effects of the mRNA vaccines. If an instance (like a doctor or an intermediary) subjectively decides whether to ignore an adverse event, this is detrimental to the statistical value of reporting.
One aspect worth considering and influencing the level of reporting is, which adverse event types seem relevant. If the purpose of a Covid vaccine is to stimulate the immune system, mild flu-like symptoms are consequential and expected rather than adverse events and one may decide not to report them. Nonetheless, flu-like symptoms are the most frequently reported adverse event types of Covid vaccines.
The standard adverse event reporting systems are VAERS for the USA and EudraVigilance for Europe.
Trajectories to a report
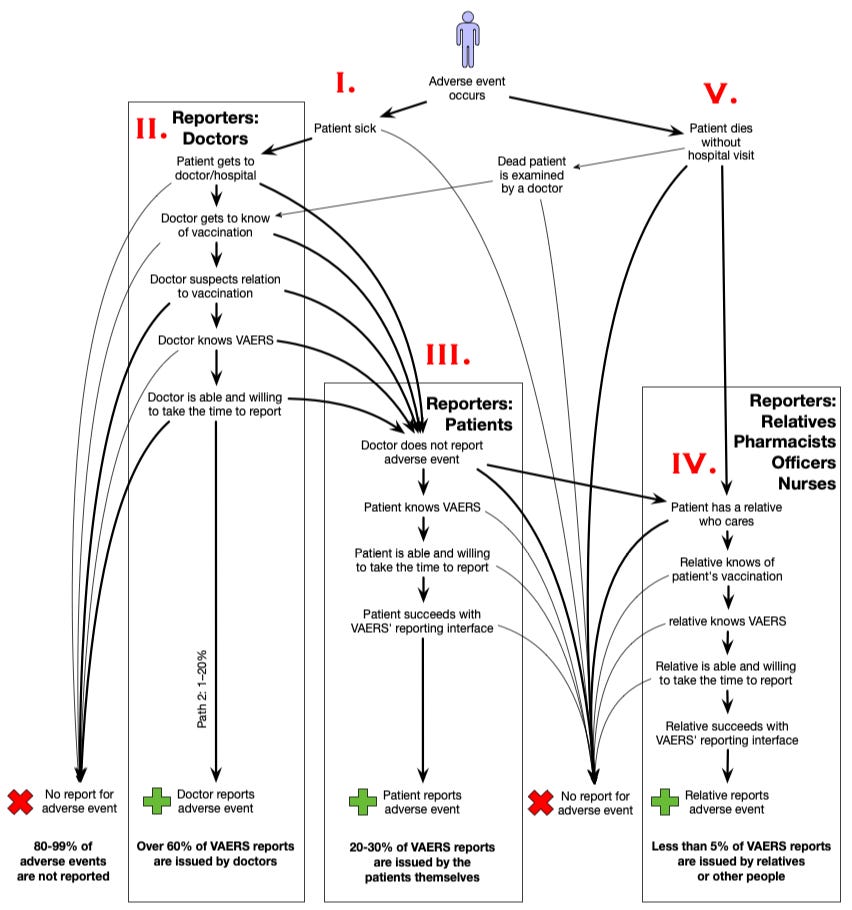
As indicated above, not each adverse event that occurs is reported to an adverse event reporting system. Many factors have an influence on the probability of an adverse event to be reported. For an adverse event, a possibly long list of conditions must be met in order to result in a report, as illustrated in the following diagram. It may look a bit complex on the first view, but I will go through its details slowly. In the beginning, at the top, an adverse event occurs. Then, different situations and conditions may occur, following the diagram arrows and developing the course of events downwards until finally, at the bottom, an adverse event report is filed – or not. Each situation or condition may have the outcome of a decision that prevents an adverse event report. So, logically, the longer the path taken (the more arrows are followed) the lower the probability, that the adverse event is reported. This is confirmed by a comparison of the percentage of reports filed by doctors, patients, or other people. The (in my opinion) main paths are drawn with bold arrows, the less frequent ones with thin ones.
I. Patient sick → go to a doctor?
If an adverse event occurs, in most cases the vaccinated person gets sick, in some cases even dies. A sick person may decide to consult a doctor or go to a hospital – or not. This is the first factor, because of which many adverse events remain unnoticed. The more severe a side effect, the more likely is the visit of a doctor or hospital, and the less probable is the adverse event to remain unreported. One diagnosed influence factor is gender: On average, 71% of the VAERS Covid-vaccine reports relate to female patients.
II. At the doctor or hospital → file a report?
The CDC is requesting doctors to report any type of adverse event – even if they are not concluding a causing relationship to vaccination. But studies reveal that, in fact, only a small fraction of the adverse events which occur, actually does get reported.
Why that? In order to file a report, a doctor needs to
know about the patient's vaccination
know an adverse event reporting system
be motivated to report any adverse event or at least suspect a relationship between symptoms and vaccination
take the conscious decision to file a report
have about 30 minutes of time per patient to enter and file the report
All but one of these factors are external and can only partially be influenced by the doctor. Factor 4, the conscious decision, is more difficult and involves motivation for and against reporting: There may exist motives which could cause a doctor to consciously decide against filing a report, despite suspecting a causing relationship:
Psychology: Repression of the idea that one's well-intended action could have caused harm
Legal: Avoiding accountability for side effects if the doctor him/herself has vaccinated the patient did not inform him/her well enough about possible side effects
Vocation: Vaccine advocacy and desire to suppress any information that could cause vaccine hesitancy
Lack of idealism: Reluctance to do half an hour of unpaid work necessary to file a report
Feeling of having done enough: Having filed dozens of adverse event reports already in the past and feeling tired of it
Frustration: Having filed dozens and having found thousands of similar adverse event reports in reporting systems and realizing that they did not have consequences
One more hindrance is that doctors may need to save time or are dependent on external confirmation by only reporting adverse events which are proven or highly probable side effects. Symptoms like anaphylaxis, thrombosis and myocarditis are obvious, confirmed, and thus expected, side effects and seem “easier” to report. More diffuse symptoms like pain in different body parts or neurological issues are hard to assign to a specific cause and more exotic symptoms like autoimmune diseases or reactivation of former viral infections are less likely to be suspected to be caused by vaccination, and thus less likely to be reported.
In summary, there is a high likelihood, that an adverse event is not reported by a doctor.
However, motivation for filing an adverse event report can be increased in some circumstances, for example by:
the motivation of doing a service to society by supporting the detection of side effects
feeling obliged to help a patient to possibly get compensated for vaccination damage
striving to take the best support possible for a friend, colleague or relative
The higher the ethical standards or scientific engagement of a doctor and the better the relationship with a given patient, the higher is the probability of a report.
III. Back home without report → File a report for oneself?
If the treating doctor did not file a report (or the patient did not ask for medical support), the patient can file a report for him/herself. For example, a PDF form can be downloaded from the VAERS website and be used to report an adverse event.
Again, there are many opposing factors: The patient needs to
suspect a relationship between vaccination and his/her symptoms
know of the existence of adverse event reporting systems
know that he/she can file reports for him/herself.
take the decision to file a report
find a communications channel to VAERS
take 30 minutes of time to provide all related data
Regarding all these, it struck me to find out that 20–25% of the Covid adverse event reports in VAERS were filed by the patients themselves!
This is a surprisingly large number: If most people with adverse events went to a doctor and most of them filed a report, there would be few ones left with the necessity to file a report themselves. Of these few ones left, only a small fraction would actually know about VAERS (maybe 5%), so the resulting quota of self-reports is rather supposed to be around 1% than over 20%. This is evidence that a majority of adverse events is not reported by doctors or that many people with adverse events do not visit a doctor (which is unlikely for severe evens).
IV. Others learn about it → File a report for s/o else?
Individuals, like relatives, colleagues, employers, caretakers or others, may directly file an adverse event report to VAERS, but face the same hindrances as the patient him/herself before. As a consequence, only a small part of the adverse events is reported this way, according to my analyses, it is less than 5% of all reported cases.
V. Sudden death
For events of sudden death after vaccination (without visiting a doctor or a hospital), a VAERS report is even more unlikely: Obviously, the deceased cannot report his/her death him/herself. The person who writes the death certificate hardly knows the dead's vaccination history if he/she did not conduct the vaccination him/herself. Only in rare cases, an autopsy is conducted. If the patient is of older age, it is less likely that anybody suspects a relation to vaccination. The deceased's bereaved have other worries than searching for vaccination documents and filing reports. If a patient dies without related persons, it is even more unlikely that somebody files a report.
Overreporting and Underreporting
On the one hand, by the nature of their definitions, there must be more adverse events than side effect events. Each side effect is an adverse event but not vice versa. In consequence, each adverse event report system contains reports of adverse events which are no side effects. This is called overreporting.
On the other hand, the diagram above shows the large number of decisions that can make an adverse event remain unreported. This is called Underreporting.
Now, which of these two effects is stronger? More importantly, what is the total result of the two effects combined? By looking at a reporting system alone, it is not possible to tell how many adverse events occurred without being reported. There are many studies that correlate VAERS data with external information, and all conclude a significant net underreporting. Underreporting factors depend on symptom and severity and estimates range from 6 in a CDC report to over 100 in a Harvard study. This means, that if 1,000 adverse events of a given type are reported to VAERS, the number of adverse events that actually occurred may be 6,000 or even up to 100,000. A British meta-study of adverse event reporting systems assembled the results of 37 different studies and calculated a median underreporting factor of 16.
Thus, claims of an overall overreporting of adverse event reporting systems like VAERS or EudraVigilance are absurd and disproved by peer-reviewed scientific studies.
In some countries, there is even one more filter step involved before a report is sent to an adverse event reporting system: Intermediary institutions, which receive the local or national adverse event reports and may reject or forward them to a reporting system. For Germany, this is the Paul Ehrlich Institut which is rumored to be quite diligent in filtering out reports. I do not know to which degree this is true, but EudraVigilance numbers raise a strong suspect: For the Netherlands, a query for Covid vaccine-related events returns 3.7 times more reports per vaccinated person than for Germany. I think that this makes it impossible to judge the statistical siginificance of an adverse event with German reporting data.
Conclusion
Many factors oppose the submission of an adverse event report. The biggest impact on reporting levels can be diagnosed to be the behavior and motivation of doctors. Overall, significant underreporting of an overall factor of around 20 is possible. From the considerations above, we should keep in mind that there are some trajectories in the creation of an adverse event reports that have special characteristics and could differ between groups of doctors, population groups and their social and professional environments.
With all these considerations, the first article’s idea of batches that are related to thousand adverse events, with only a few of them actually reported, gets a little less utopian. These are some interesting aspects to research in the third and last article of this series: The Real Story behind the ‘Bad Batches’.
Stay tuned, stay critical and keep thinking for yourself,
Leonard Frey
(Pseudonym)




https://public.tableau.com/app/profile/alberto.benavidez/viz/WelcomeTheEaglesVAERSDashboard/ByLotLookup
I think you will appreciate by public Tableau VAERS Interactive Dashboard, especially the visuals that measure the "lag days" between vaers receive date and publish date. I think people assume that VAERS publishes everything the receive? Minus of course the duplicates and false claims that do not make it into the public domain. They do have up to 4-6weeks to publish a report, yet there is also 5000+ reports that have been deleted even after being made public. Fair enough, however I can only find maybe 10% of the other half of the duplicate claim. How big is there "to be processed" inventory of reports? Did you know there have been thousands (at least ~20K reports) that have been made public 150 days after received? Many more thousands if you look at reports made public 90days (3months) after they received claim. More to follow...